The chart below shows the Nationwide average price of gasoline over the last year (12 months) compared to the gas price in Dallas and the price in Los Angeles. You can change the time period to see anywhere from 1 month to 6 years. Simply select the time period you wish to see from the list at the top. You can also choose different cities and time periods from the customization box below the chart.
| Richmond Historical Gas Price Charts Provided by GasBuddy.com |
The following table compares the current average gas price statewide between New York, California and Texas. As of this writing, the result is as you would expect… Texas gas prices are lower than New York and much lower than California.
At first glance, you might think that it is purely a function of the proximity of oil wells. After all, Texas is known for its oil wells. However, after a bit of reflection you might think refinery location would actually be more important. After all, if oil has to be shipped long distances to a refinery and then the gas has to be shipped back, it isn’t going to help the local price of gas.
| Gas Prices around the country |
 Yes, Texas has 28 refineries, but California has 17 refineries, and New York doesn’t have any. Although New Jersey has 6 (although 3 of them have been closed) and Pennsylvania has 6 more that are operating. Therefore distance to refineries is not a significant factor for any of the three states we are looking at. For more information see Complete List of U.S. Oil Refineries.
Yes, Texas has 28 refineries, but California has 17 refineries, and New York doesn’t have any. Although New Jersey has 6 (although 3 of them have been closed) and Pennsylvania has 6 more that are operating. Therefore distance to refineries is not a significant factor for any of the three states we are looking at. For more information see Complete List of U.S. Oil Refineries.
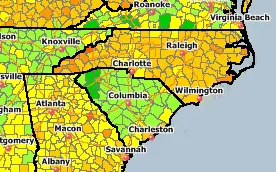
Plus, when we look at the list of Oil Refineries in the U.S. neither North or South Carolina have any refineries. And as we can see on the Gas Price Chart South Carolina has significantly lower gasoline prices than North Carolina and even adjacent counties across state lines have significant price differences.
So why would prices differ so drastically across state lines?
Obviously, the key factor in gas prices is not the distance to a refinery…
But some arbitrary line on a map. Hmmm, could it be related more to governments than to oil companies?
When we look closely we see that the amount of state sales and highway taxes included in the price of gasoline varies greatly and is the key factor to the price differential. According to the American Petroleum Institute : (as of 1/15/16) (follow the link to API for the most current rates).
| State | State Taxes | Federal Excise Tax | Total Taxes per Gallon |
| Texas | 20¢/gal. | 18.40¢/gal. | 38.40¢/gal. |
| New York | 42.64¢/gal. | 18.40¢/gal. | 61.04¢/gal. |
| California | 40.62¢/gal. | 18.40¢/gal. | 59.02¢/gal. |
And if we we look at the differences in taxes between North and South Carolina we see:
| State | State Taxes | Federal Excise Tax | Total Taxes per Gallon |
| North Carolina | 35.25¢/gal. | 18.40¢/gal. | 53.65¢/gal. |
| South Carolina | 16.75¢/gal. | 18.40¢/gal. | 35.15¢/gal. |
So it is obvious why people from North Carolina drive across the state line to get gas (to save 18.5¢/gal. in taxes).
You might also like:
- Inflation Adjusted Gasoline Prices
- Gasoline vs. Crude Oil Prices
- Historical Gas Price Chart
- Current Average Cost of Gas by County
- Gasoline Taxes by State
- Average Cost of Gas Per Month
- Calculating Miles Per Gallon
- Regarding Gas Inflation
- Historical Oil Prices Chart
- Gasoline Rewards Credit Cards
- More Inflation Adjusted Prices


I’m finding your Canadian prices to be higher than, for example, when I query on GasBuddy.com, itself. For example, just for comparison’s sakes, if I run a query on that sight, in it’s fuelinsights section (http://fuelinsights.gasbuddy.com/Charts), comparing U.S. National and Canadian National Regular gasoline price, Canada (on Feb. 15, 2017) is at $3.053 (USD). You’re showing it at something near $4.04 (can’t see the data to get the exact amount).
Can you tell me if there’s another variable that’s not obvious? It looks like you’re both claiming USD currency. Inflation (current vs. some historic value)?
Thanks!
Pete,
Not sure why their data would be different since our data comes from a direct feed from GasBuddy we actually have no control over it. The only part we control is the table.
your state+federal tax numbers in the chart is adding something twice – CA total with federal is not $.89/gal – it’s more like $.69 so I think you’re adding the .18 federal twice in your calcs.
Lyle,
Thanks for pointing that out! Note that several states have actually decreased their gasoline taxes since the article was originally published back in 2013 (perhaps due to the decrease in oil prices). I’ve updated the hard-coded numbers in the tables as of January 15, 2016. If you follow the link http://www.api.org/oil-and-natural-gas-overview/industry-economics/fuel-taxes/gasoline-tax embedded in the article you will get the current rates. If you click on California on that map it pulls up a PDF document telling about the decrease in gasoline taxes and a corresponding increase in Diesel taxes.
Taxes is one of the main reasons Oklahoma has the worse roads in the nation but the lowest fuel taxes. You get what you pay for.
Except when Government and corruption are involved then you always get less than you paid for. 🙂
So you would agree that zero taxes would make those Oklahoma roads the best in the country?
Not sure how you came to that conclusion?